Feasibility, acceptability and costs of nurse-led Alpha-Stim cranial electrostimulation to treat anxiety and depression in university students
Royal, S., Keeling, S., Kelsall, N., Price, L., Fordham, R., Xydopoulos, G., et al. Feasibility, acceptability and costs of nurse-led Alpha-Stim cranial electrostimulation to treat anxiety and depression in university students. BMC Primary Care. 2022; 23:97.
Funding Source, Location of Study or Author’s Affiliation
University of Nottingham Health Service, Cripps Health Centre, United Kingdom. Funded by National Institute of Health Research (NIHR), with the devices being loaned to the clinic by the manufacturer.
Device
Alpha-Stim® AID
Key Variables
Anxiety and Depression.
Objective
To evaluate the effectiveness and the cost effectiveness of a new medical device for the treatment of mild to moderate depression and anxiety in Primary Care when delivered through an innovative nurse-led clinic. Assess a group of participants attending the mental health clinic who used Alpha-Stim AID either alone or in combination with other treatments. Ispero was used to gather a baseline PHQ-9 and GAD-7 scores and then compared this with a score taken after 8 (6-10) weeks of use.
Design
- The study evaluated the effectiveness of a new treatment without increasing the cost burden. It was conducted at a GP surgery in the City of Nottingham and ran for six months.
- The first study procedure was registration with the Ispero system (CE-marked but not currently part of standard management) and completion of the baseline assessments it contains. The results of this will be available immediately and will assist the clinician in identifying the most appropriate management option.
- Management options include:
- Self help
- Support agencies including NHS, University, and voluntary sector.
- Psychological therapy with one of the three NHS provider organizations in Nottingham.
- Alpha-Stim treatment for those with generalized anxiety (CE-marked but not currently standard management).
- Referral to a GP to discuss medication.
- The participants responses were monitored by Ispero and used at future contacts to guide further management.
- After eight weeks an investigator contacted the participant to ask permission to inspect their Ispero data and collect outcome measures. They were then offered an opportunity to withdraw consent for participation. In addition, participants were offered a personal appointment with a study clinician so that data can be reviewed in a consultation environment.
- Participants completed a short user satisfaction survey.
- A control group of patients attending their GP practice with mental health problems that would have been eligible for inclusion in the study were identified at the end of the study and a retrospective notes review will be performed by the PI.
- Study design has been chosen as it aligns with current NHS priorities and causes minimal disruption to participants and local services.
- A non-randomized controlled trial was chosen as it seemed more ethical than a randomized control trial. In addition, it would create logistical problems that would not outweigh the benefits in outcome data quality and comparability.
Primary Outcome Measures
To compare PHQ-9, GAD-7 and satisfaction survey responses in the experimental group with a selection of patients receiving usual care in an ad-hoc control group.
Secondary Outcome Measures
- To determine the effectiveness of the new National Health Service (NHS) pathway (Alpha-Stim CES) in managing the conditions when compared with a group of patients receiving standard care and establish its acceptability to patients and providers.
- To compare the cost-effectiveness of the new NHS pathway with usual care.
- Explore levels of service use and use of prescription medication.
Key Inclusion Criteria
- Adults with mild to moderate anxiety and/or depression symptoms presenting to primary care.
- Be registered with the practice.
- Aged 18 or over.
- Adult patients attending the participating practice requesting an appointment to discuss a mental health problem.
- Not requiring immediate referral to secondary care because of symptom severity.
- Capacity to understand the patient information sheet and give consent to participation.
Key Exclusion Criteria
- Patients not suitable for the nurse-led mental health clinic would include those who appear distressed or at acute risk of harm to themselves or others or who appear to be intoxicated.
- Patients with mental health problems related to a terminal or acute physical illness.
- Unable to communicate effectively in English.
Protocol Summary
- Entry into the study were offered to attendees at a walk-in clinic for people who have mental health problems.
- The clinic is located at the student health center on the University of Nottingham main campus.
- All attendees were assessed by a trained mental health nurse on presentation to the service.
- Non eligible patients were offered usual care according to the standard operating procedures of the service.
- Patients who were eligible to join the study were given an information sheet and time to consider participation.
- Patients who did not give consent were offered usual care according to the standard operating procedures of the service.
- Patients who consented were entered into the trial.
Device Application Protocol
Alpha-stim® AID is small battery powered device that passes microcurrents through the brain of the user to stimulate alpha waves. The treatment is delivered by two small clips attached to each ear and lasts for between twenty minutes and one hour each day. This technology has been used as a treatment for anxiety and depression for over forty years and has a large supporting scientific literature including a recent systematic review. Electrical stimulation was applied via ear clip electrodes at the lowest setting (0.5 Hz, 100 μA) based on previous positive studies of the device for depression and anxiety. Study Duration: Over 6-10 weeks, with measurements taken at 8 weeks. No adverse events were reported during the trial and nine patients expressed a high degree of satisfaction with their Alpha-Stim treatment in solicited feedback collected at the end of the study. No negative feedback was received.
Statistical Analysis Plan
- Any data extracted for comparison was fully anonymized and no patient identifiable data was seen by any person who is not an employee of the study practice. The null hypothesis was that the new pathway is no more effective than standard treatment.
- Compare the scores with those from an equal number of young people who did not enter the treatment pathway and were seen by a doctor in the first instance (usual care).
- As part of routine service evaluation, PHQ-9 and GAD7 scores were collected between six and ten weeks of treatment following first presentation and the data was used as an ad-hoc comparator.
- Scores were collected over the same period (January to May 2020) and came from patients with new presentations of minor mental health problems which would have been suitable for the pathway had space on it been available.
Results
Subjects
- Active Cohort
- An initial sample of 48 participants consented to the study and were initially loaned units.
- 47 young people (mean age 21.4 years, 77% female) contributed data to active part of the study.
- One patient returned their Alpha-Stim® as they had not disclosed the use of illegal stimulants and decided to seek alternative treatment.
- One patient person broke their device accidently after four weeks but did contribute scores to the study.
- Comparator Control
- A sample of 47 control group scores were identified by an investigator who was not part of the trial (NK).
- The mean age of this group was 22.7 years and 79% were female.
- Follow Up
- There were 187 contacts during a total of 2,667 days of supervised management in the intervention group which is equivalent to contact every 14.3 days.
- In the control group there were 156 contacts in 2,392 days which is equivalent to a contact every 15.3 days.
Data Analysis
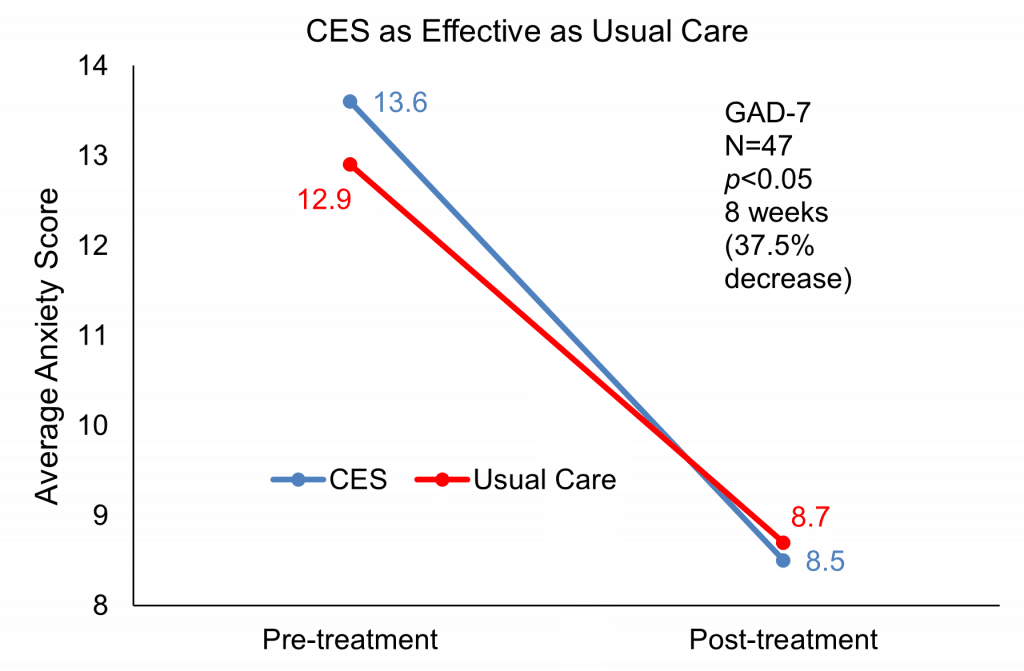
Intention to treat and completed group analyses was performed for the behavioral measures. The mean baseline in anxiety was a GAD7 score of 13.6 and the mean score after treatment was 8.5. This improvement was significant at p<0.05. In the control group, the mean baseline GAD7 score was 12.9 and the mean post-treatment score was 8.7, a significant (p<.05) decrease. These results show that Alpha-Stim CES is as effective as the NHS usual treatment pathways for GAD.
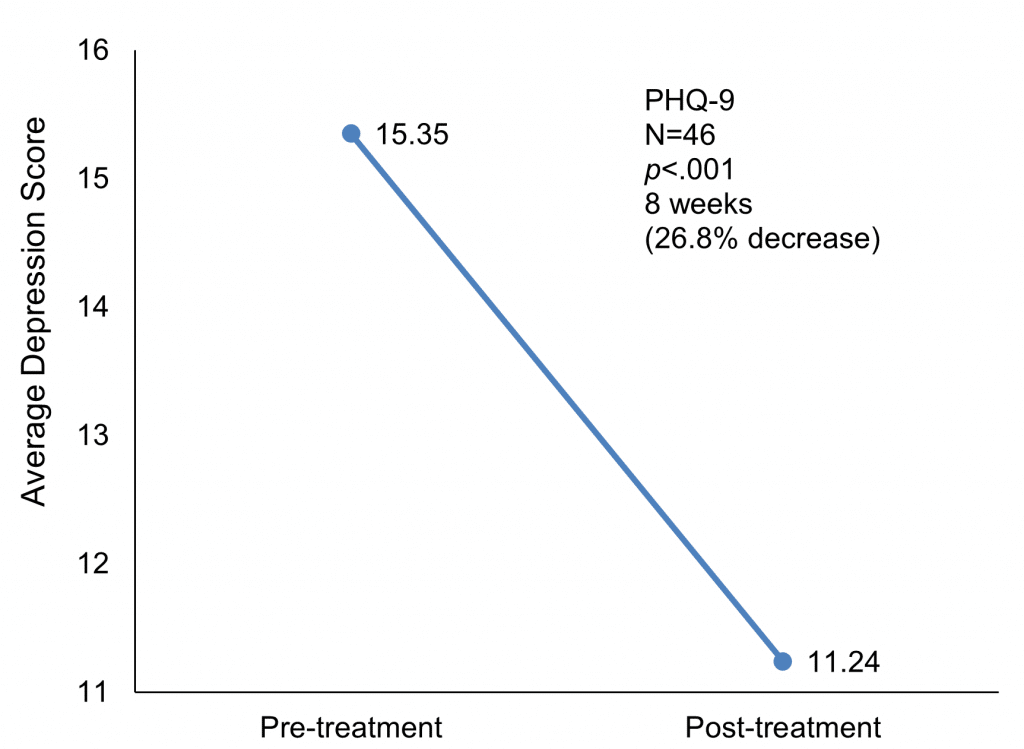
The mean baseline in depression was a PHQ-9 score of 15.35 and the mean score after treatment was 11.24 – representing an improvement that was significant at p<0.001. No PHQ-9 scores are reported for the control group.
Conclusions
The results of this study suggest that a care pathway incorporating Alpha-Stim was non-inferior to usual care and may indeed be slightly better in terms of mean reduction of PHQ-9 and GAD-7 scores.
The researchers estimated that a 20-minute appointment in the nurse-led clinic costs £23.67 ($30.41) and that a 20-minute appointment with a doctor as part of usual care calculated by the same formula is £42.61 ($54.75).
Given the similar rate of consultation the authors believe the Alpha-Stim pathway represents a significant cost saving likely to be in the region of 10-20% allowing for fewer and shorter consultations in the usual care group.
No adverse events were reported during the trial and nine patients expressed a high degree of satisfaction with their Alpha-Stim treatment in solicited feedback at the end of the study. No negative feedback was received.
Limitations
Given this study included a sample of patients presenting to primary care, participants were not able to be randomly assigned to CES or usual care groups.
Study Quality: GOOD