Treatment of Sleep Disorder with Cranial Electrotherapy Stimulation Combined with Psychological Counseling in Postmenopausal Women: A Randomized Study
Kong, J., Wu, J., Jiang, X., et al. Treatment of Sleep Disorder with Cranial Electrotherapy Stimulation Combined with Psychological Counseling in Postmenopausal Women: A Randomized Study. Chinese Journal of Medicinal Guide. 2014; 921, 16(6):(Serial No.128).
Funding Source, Location of Study, or Author’s Affiliation
Women Health Care, Nanjing Maternity and Child Health Care Hospital, Nanjing, China
Device
Alpha-Stim®
Key Variables
Menopausal symptoms, sleep, depression, and anxiety.
Objective
To compare the effects of Cranial Electrotherapy Stimulation (CES) combined with psychological counseling for the treatment of disordered sleep in post-menopausal women.
Design
Investigate the occurrence of insomnia and its frequently secondary impact on depression, anxiety, and quality of life in peri- and post-menopausal women. Assess the efficacy of CES, a non-pharmacological FDA cleared treatment for insomnia in conjunction with psychological counseling and compare it to psychological counselling alone. Measure changes in symptom profile at baseline, 4, and 8 weeks after treatment.
Primary Outcome Measure
- Menopausal symptoms as measured by the Kupperman Index (KI) based on the grade of 0-3. The KI items include
- Vasomotor symptoms (hectic fever and sweating)
- Sensory disturbance
- Insomnia
- Depression
- Dizziness
- Fatigue
- Joint and muscle pain
- Headache
- Palpitation
- Skin formication
- Hyposexuality
- Urinary tract infection
- Sleep as measured by the Pittsburg Sleep Quality Index (PSQI) based on the grading score of 0-3 on the 18 scoring items divided into the 7 components of sleep:
- Quality
- Latency
- Duration
- Efficiency
- Disturbance
- Daytime dysfunction
- Medication use
Secondary Outcome Measure
- Anxiety indicated by the Self-Rating Anxiety Scale (SAS) based on the grade scoring of 1-4
- Depression assessed by the Self-Rating Depression Scale (SDS) based on the scoring grade of 1-4
Key Inclusion Criteria
- Pittsburgh Sleep Quality Index (PSQI) score ≥7
- Women aged ≥40 and ≤60 years old amenorrhea for ≥3 and ≤12 months
- Perimenopausal symptoms as indicated by a score of ≥15 in the modified KI
- Aged ≥40 years old with inconsistent menstrual cycles not attributed to pregnancy
- Follicle Stimulating Hormone (FSH) levels ≥10U/L
- Women who have undergone artificial menopause or ovarian ablation ≥ 6 weeks with FSH ≥10U/L
- Intact uterus and bilateral ovaries
Key Exclusion Criteria
- Non-perimenopausal women
- Sex hormone treatment in preceding three months
- Diagnosis of
- Hyperthyroidism
- Coronary atherosclerotic heart disease
- Hypertension or pheochromocytoma
- Neurasthenia
- Psychosis
- Other diseases closely associated with primary symptoms
- Taking medication for insomnia, anxiety, or depression
- Men
Protocol Summary
A randomized Intent-to-Treat (ITT) study of women aged 35-40 years presenting with disordered sleep and meeting full eligibility criteria were enrolled in the study. A total of 139 women were randomly allocated into two groups. Group A received psychological counseling and CES (CES+PSY). Group B received weekly psychological counseling (PSY). Peri- and post-menopausal symptoms were assessed following the Technical Guidelines for Clinical Research on Treatment of Menopausal Syndrome with Traditional Chinese Medicine (TCM) and Natural Medicines issued by the China Food and Drug Administration. FSH levels were taken on the 3rd day of menstruation for peri-menopausal women. Women in the PSY + CES condition received treatment using the Alpha-Stim daily for 60 minutes over eight weeks and psychological counseling once a week. Patients receive supervision for the first three treatment sessions and then self-administered CES at home for the remainder of the study. The comparator group received psychological counseling once a week for eight weeks.
Device Application Protocol
Alpha-Stim was used to administer CES to Group A (CES+PSY). Patients were instructed to turn on the power, coat the bilateral earclips with conductive fluid and attach them to each earlobe. During self-administration, patients were instructed to gradually increase the current intensity from 10-500 μA, and to calibrate the maximum dosage at a level that was comfortable for them or when they felt a tingling sensation. The duration of each treatment cycle was 60 minutes and occurred daily over eight weeks.
Statistical Analysis Plan
Using prevalence rates for insomnia among peri- and post-menopausal women, a power calculation estimated a sample size of 60 cases, with the margin for a withdrawal rate of 20%, the total cases were determined as 72. The ratio of the experimental group to the control group was 1:1, and the random coding table followed the order in which the cases were selected. Software was used to obtain random numbers and to generate random grouping. Data analysis was conducted using SPSS 17.0 and Excel 2010. Statistical analysis included t-test, chi-square test, and analysis of variance (ANOVA). Higher scores are indicative of greater impairment (e.g., high KI indicative of multiple menopausal symptoms or high PSQI score suggestive of poorer sleep quality) and taken at baseline, followed by 4- and 8-weeks post-treatment.
Results
Subjects
A total of 139 peri- and post-menopausal women aged 35-40 years were recruited to the study as they attended the Nanjing Maternity and Child Health Care Hospital, in China. The rate of subject attrition was 12.2% (N=17), with eight patients lost to follow up or withdrawn from the study before or at baseline, resulting in 131 patients considered in the ITT analysis. A further nine patients withdrew from during treatment, resulting in 122 patients completing the 8-week study, with 63 cases in Group A and 59 cases in Group B. The mean (SD) age of Group A was 47±4 years and 46±4 years in Group B. The difference in age distribution between conditions was not statistically significant (p=0.52). The difference in marital status, education level, etc. was also not statistically significant (p=0.38, p=0.20).
Data Analysis
Menopausal Symptoms: The KI scores at baseline between the two groups were not significantly different (p=0.67) with a mean (SD) of 22.5±7.1 for the CES+PSY group and 23.3±6.2 for the PSY group. The reduction in KI scores in the CES+PSY group after four weeks was more than 7, suggesting an improvement in peri-menopausal symptoms, and after 8 weeks, the reduction was 12.4, suggesting a greater reduction in symptoms with prolonged CES treatment. In comparison, for the PSY group, at 4 weeks the reduction in KI scores was 4 and 5.6 at 8 weeks (p=0.00). Further details are shown in the following table.

Comparison of mean and standard deviation of peri-menopausal symptoms and sleep quality before and after treatment in both groups.
Anxiety & Depression Symptoms: Patients in both groups showed symptoms of moderate to severe depression and anxiety at baseline, but no significant difference between the groups was reported (p>0.05). After eight weeks of psychological counseling and the addition of CES in Group A, depression and anxiety symptoms of both groups were significantly lower, with a symptom reduction rate of over 50%. The mean improvement rate of depression and anxiety symptoms in CES+PSY was 7%-10% higher than that of PSY the group, a statistically significant difference (p=0.00).
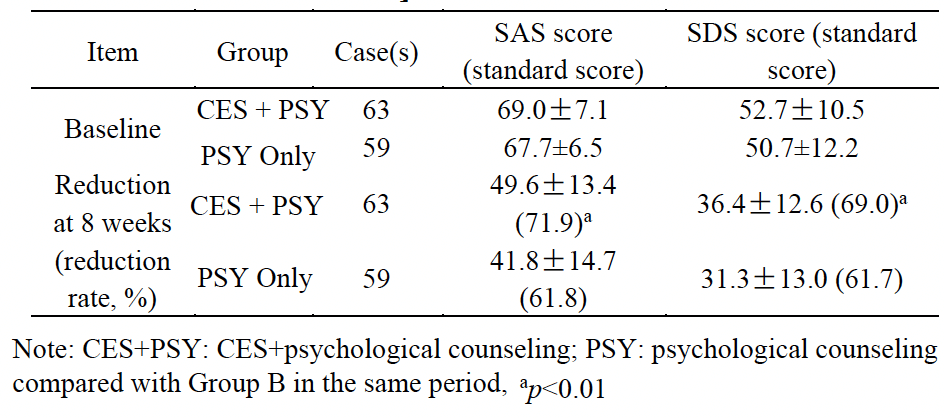
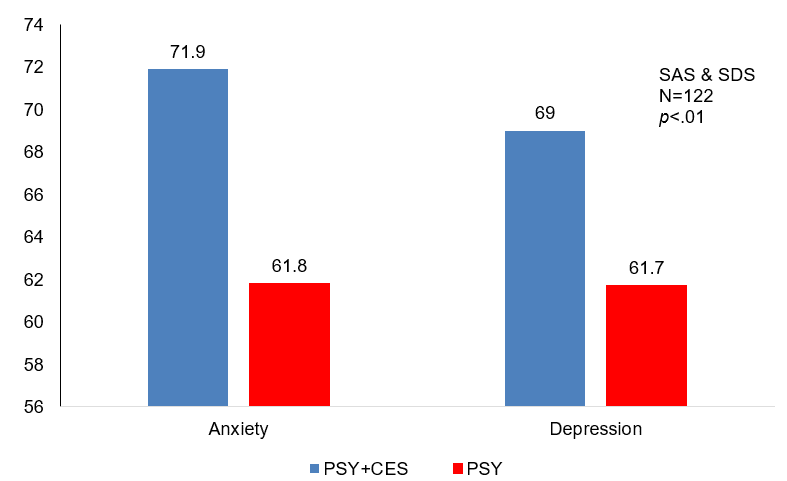
Percent improvement in anxiety and depression
Sleep Disruption: The PQSI scores at baseline were not significantly different (p=0.09), Group A’s mean (SD) score was 15.1±2.5 and 14.5±3.0 for Group B. After 4 weeks after treatment, the reduction of Group A was more than 7, suggesting an improvement in insomnia, with greater CES treatment resulting in greater symptom relief as evidence by a further reduction of more than 9 points at 8 weeks post-treatment. The table above shows the comparison to Group B, with a score reduction of 3.7 at four weeks and 6 at eight weeks, which were significantly less than the score reductions seen in Group A (p=0.00).
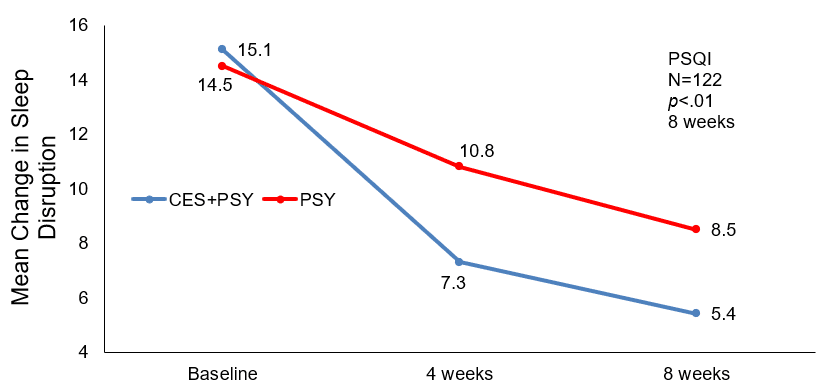
Mean Change in Total PSQI score
Safety Analysis: Group A reported six cases of mild and controllable adverse reactions. The common side-effects included dizziness, nausea, and skin-tingling sensation, and ceased immediately after reducing the intensity or stopping CES treatment. These reported reactions are expected sensations when current is turned too high and are addressed in the published instructions for using Alpha-Stim. Group B reported no adverse reactions.
Conclusion
The main treatment approach for menopausal symptoms is Hormone Replacement Therapy (HRT). Although effective, HRT has a relatively small therapeutic dose range, so adverse reactions may easily occur. HRT is not suitable for all patients, including those with malignant tumors associated with sex hormones, such as ovarian and uterine cancer. In some situations, HRT may increase the risk of ovarian cancer or endometrial cancer. CES treatment offers a non-drug approach for the management of perimenopausal syndrome that is effective in reducing menopausal symptoms, sleep disturbance, and emotional distress. Furthermore, it is safe, with extremely low risks for any patient.
This study demonstrated CES combined with psychological counseling resulted in faster treatment response and significant improvements to menopausal symptoms (p<0.05) and reduction in the occurrence of disordered sleep (p<0.01). In addition, compliance rates as evidence by lost-to-follow-up were lower in the CES condition. The authors conclude that CES avoids the possible adverse reactions common in medication for the management of insomnia and warrants further clinical investigation. The authors proposed a mechanism of action where CES promotes the secretion and balance of 5-hydroxytryptamine, dopamine, norepinephrine, and other neurotransmitters in the brain and improves the imbalance of neurotransmitters caused by the decline of estrogen level.
Limitations
The authors point out the following limitations in the study. The observation period was only eight weeks, the longer-term therapeutic efficacy needs to be studied. Sleep was evaluated only by a self-report measure, a more in-depth study assessing sleep structure (such as polysomnography) should be conducted.
Study Quality: FAIR

